Introduction to Food Microbiology and Safety
The Unique Metabolic Characteristics of Lactic Acid Bacteria
Welcome to our in-depth exploration of the unique metabolic traits that set lactic acid bacteria apart from other Gram-positive bacteria. In this article, we’ll uncover why lactic acid bacteria lack catalase and instead use NAD peroxidase to thrive in environments both with and without oxygen. Let’s dive into the fascinating metabolic mechanisms that make lactic acid bacteria ideal for various food fermentation processes.
The Unique Fermenting Abilities of Lactic Acid Bacteria
To discuss why lactic acid bacteria (LAB) do not possess catalase, we must first understand the distinctive metabolic characteristics of these bacteria, which predominantly engage in lactic acid fermentation.
Generally, when oxygen is present, most microorganisms break down sugars into carbon dioxide and water through the Krebs cycle (also known as the citric acid cycle or TCA cycle). However, in the absence of oxygen, they rely on glycolysis for energy, producing by-products like acetic acid and lactic acid.
One of the defining features of lactic acid bacteria is that they exclusively undergo lactic acid fermentation, regardless of oxygen availability. This sets them apart as they do not rely on the electron transport chain or Krebs cycle for energy production. Instead, their growth is fueled by fermentation alone, which is quite unique among Gram-positive bacteria.
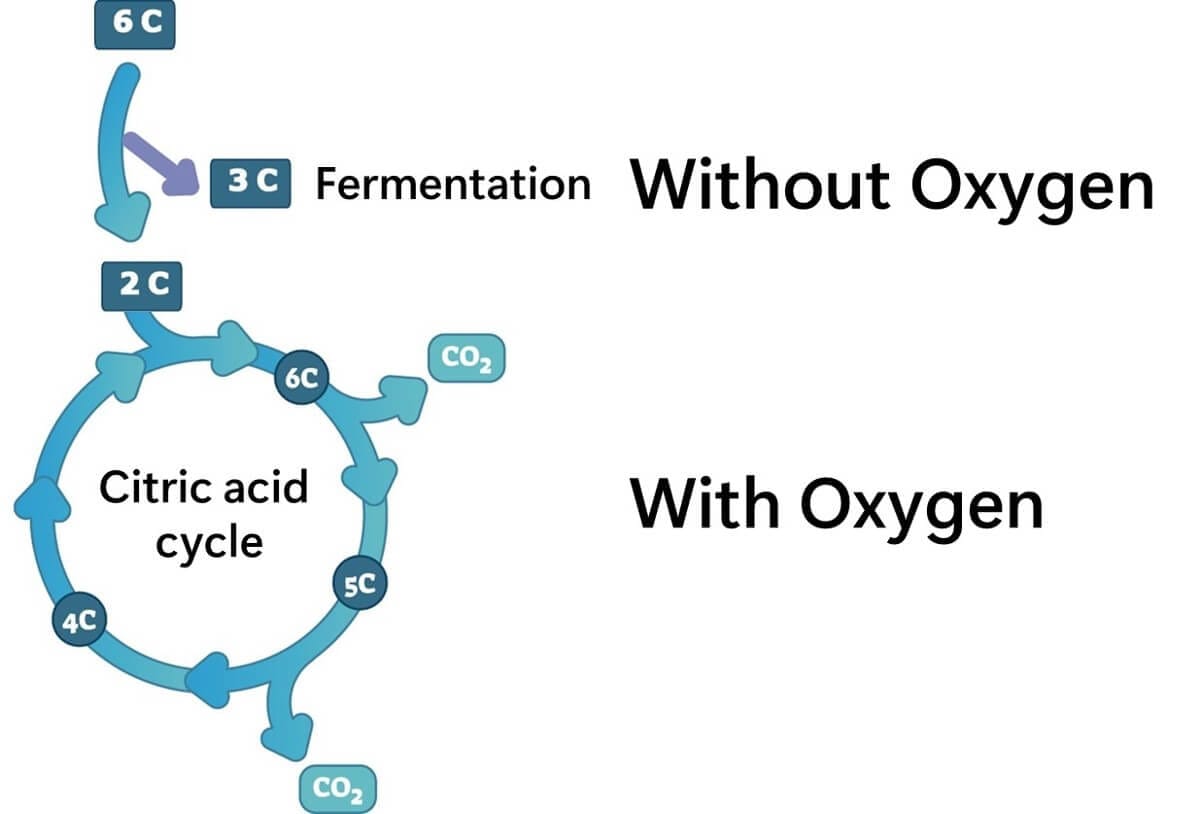
It should be noted that certain lactic acid bacteria have the genes necessary for electron transport chain enzymes (Duwat et al., 2001). In rare cases, when an external source of heme (which they cannot synthesize themselves) is provided, respiratory activity via the Krebs cycle has been observed under oxygen-rich conditions (Pedersen et al.,2011). However, this does not represent a typical metabolic trait of lactic acid bacteria and occurs only under specific conditions.
For a more comprehensive explanation of the fundamental concepts behind microbial fermentation and respiration, I recommend referring to the blow article in this blog, which provides a clear overview.
Fermentation Metabolism vs. Krebs Cycle
Fermentation metabolism offers lactic acid bacteria a significant advantage, especially in the production of fermented foods. Their unique ability to rely exclusively on lactic acid fermentation means they can perform consistently in both oxygen-rich and oxygen-poor environments.
To draw an analogy, think of lactic acid bacteria as reliable employees in the fermentation food industry. Much like a diligent worker who consistently produces high-quality results, lactic acid bacteria provide reliable fermentation regardless of environmental conditions. This consistency makes them invaluable in industries that depend on lactic acid production.

In contrast, other facultative anaerobes change their metabolic pathways depending on oxygen levels, resulting in varied performance. These bacteria are like employees with inconsistent work habits, producing different metabolites based on their surroundings, which can complicate food production.
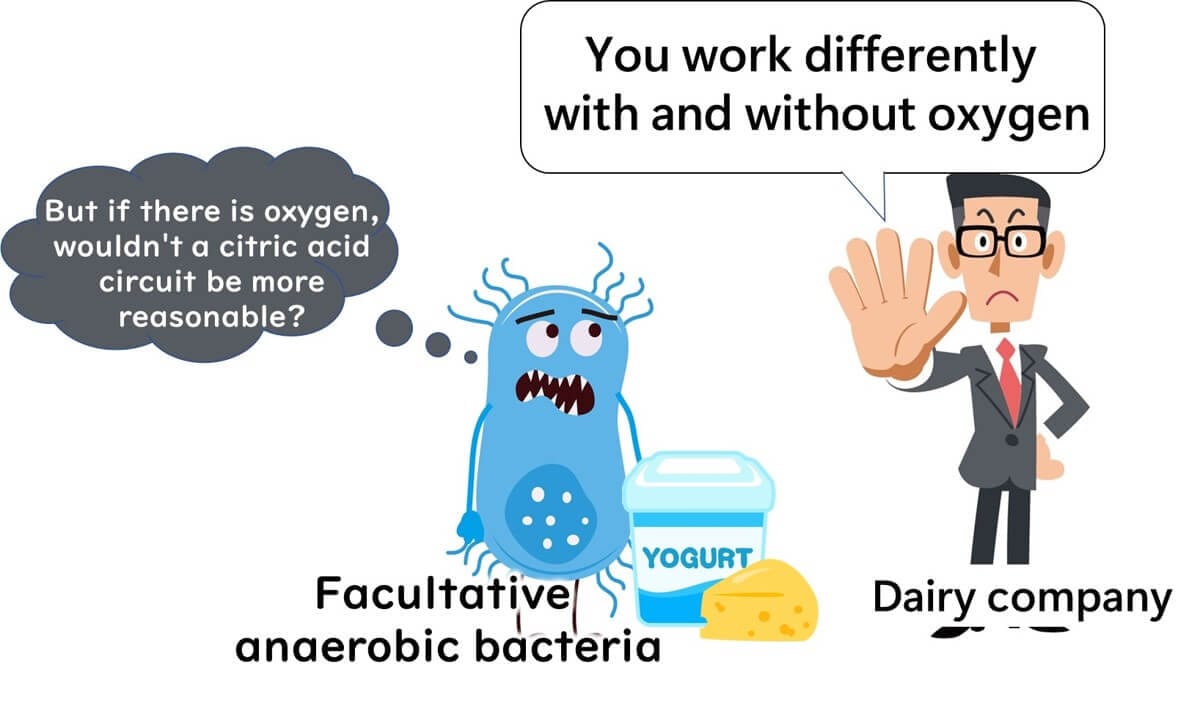
If lactic acid bacteria were to alternate between fermentation and respiration based on oxygen availability, their lactic acid production would become inconsistent, rendering them unsuitable for fermented foods.
Why Do Lactic Acid Bacteria Only Perform Lactic Acid Fermentation? - A Biological Mystery
It may seem counterintuitive, as lactic acid bacteria could theoretically generate more energy by utilizing the citric acid cycle in the presence of oxygen. However, they opt for lactic acid fermentation regardless, raising intriguing biological questions.
To understand this, we can look at a concept known as overflow metabolism. Constructing the citric acid cycle is labor-intensive, requiring a significant number of enzymes. While this cycle yields a high energy output, the cost of enzyme synthesis is substantial.
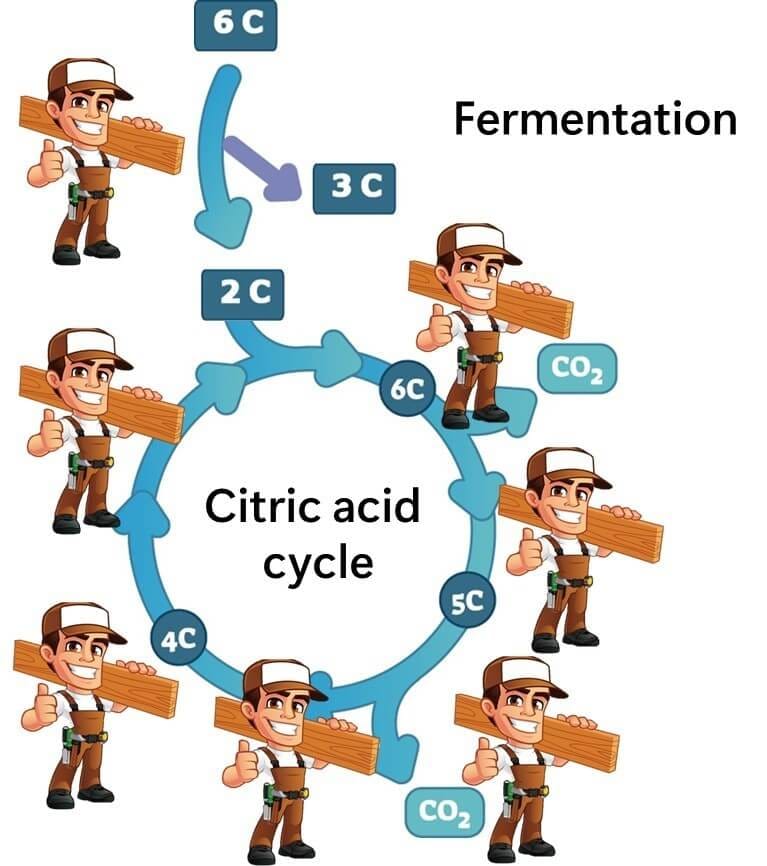
Fermentation, on the other hand, is relatively straightforward and requires fewer enzymes, albeit producing less energy overall. Even bacteria like E. coli sometimes choose fermentation when a rapid energy supply is needed in glucose-rich environments, despite the availability of oxygen (Basan et al., 2015).
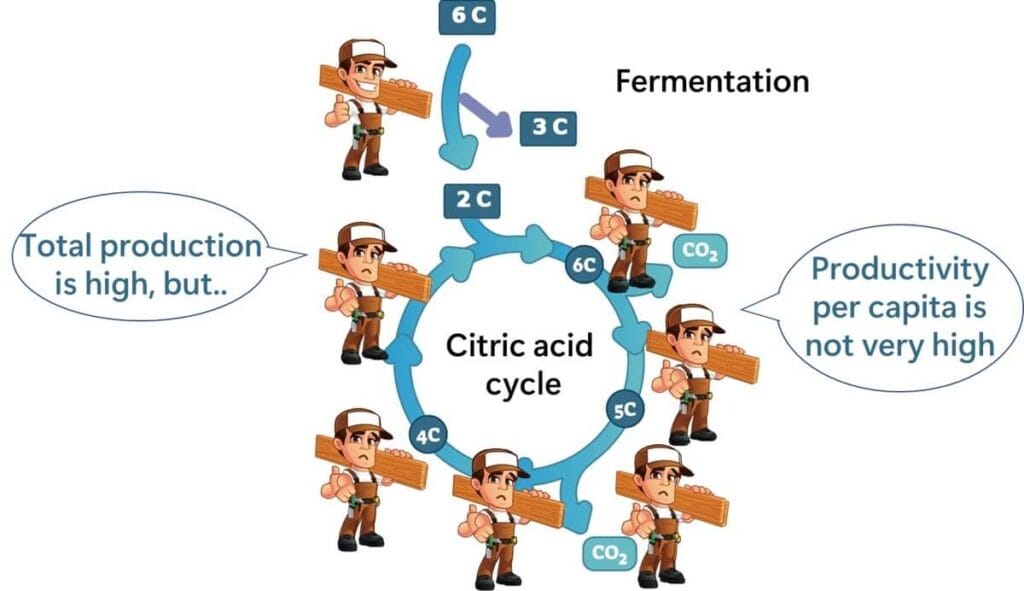
Take a look at the following graph, which plots total enzyme protein levels against ATP production. The graph demonstrates that, in terms of ATP produced per enzyme, fermentation is more efficient than the citric acid cycle, even if the latter produces a higher overall energy yield.
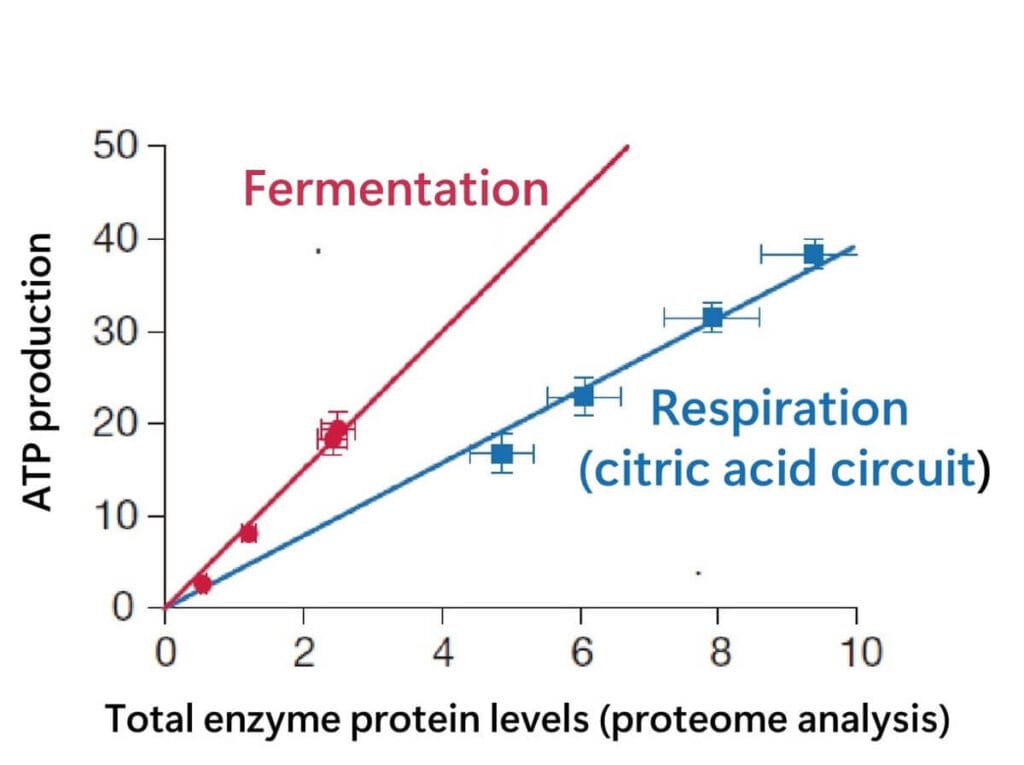
The figure above was drawn by the author from data in the following literature.
In simple terms, while fermentation doesn’t produce as much total energy, it allows lactic acid bacteria to be metabolically efficient with fewer resources, making it a practical and effective choice.
Conclusion
By understanding the unique metabolic characteristics of lactic acid bacteria, we gain insight into why they are so widely used in food fermentation processes. Their preference for lactic acid fermentation, regardless of oxygen availability, makes them an essential part of the food industry. In the next article, we will delve into the classification and diverse applications of lactic acid bacteria, particularly in food preservation and flavoring.
Related posts:
Using the Catalase Test to Identify Lactic Acid Bacteria among Gram-Positive Microorganisms (3) Hello everyone! In this article, we’ll uncover a fascinating aspect...

Discover expert-led lessons in food microbiology designed for professionals and beginners alike.

Author of this Blog: Bon Kimura
Bon Kimura, Professor Emeritus at Tokyo University of Marine Science and Technology (TUMSAT), specializes in food microbiology. He obtained his PhD from Kyoto University and became a professor at TUMSAT in 2006, serving as Dean of the Faculty of Marine Science from 2012-2015. Kimura has published over 200 international papers on food safety, pathogens, and spoilage bacteria. He has received multiple awards, including the Japanese Society for Food Microbiology Award (2019). Kimura also served as an editor for the International Journal of Food Microbiology from 2012 to 2024, where he was the principal reviewer for 1,927 papers.

Copyright © Introduction to Food Microbiology and Safety All Rights Reserved.