Introduction to Food Microbiology and Safety
The Importance of Environmental Monitoring in Food Factory Hygiene Management
In the context of food factory hygiene management, the Environmental Monitoring Programme (EMP) plays a critical role as a tool for microbiological testing. Numerous cases of foodborne illness and product recalls have shown that the implementation of HACCP alone is not sufficient to prevent microbiological contamination. This article outlines the fundamental principles of environmental microbiological monitoring in food manufacturing plants.

Why is Environmental Monitoring Essential in Food Hygiene Management?
Despite the introduction of HACCP systems in food factories, many global foodborne illness cases stem from deficiencies in prerequisite programmes. These deficiencies typically relate to inadequate validation and verification of sanitation (including facility hygiene and design) and GMP (Good Manufacturing Practices, including hygiene zoning).
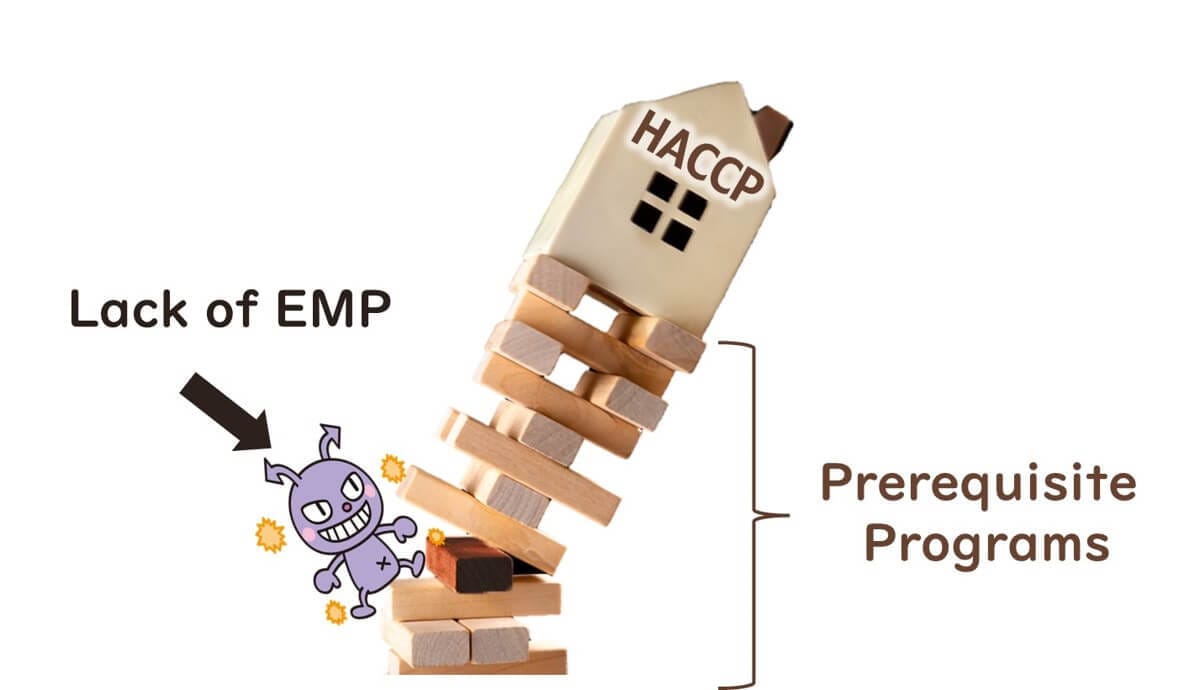
This is particularly evident in foods that are packaged after heat treatment (e.g., ready-to-eat products), where secondary microbial contamination frequently occurs.
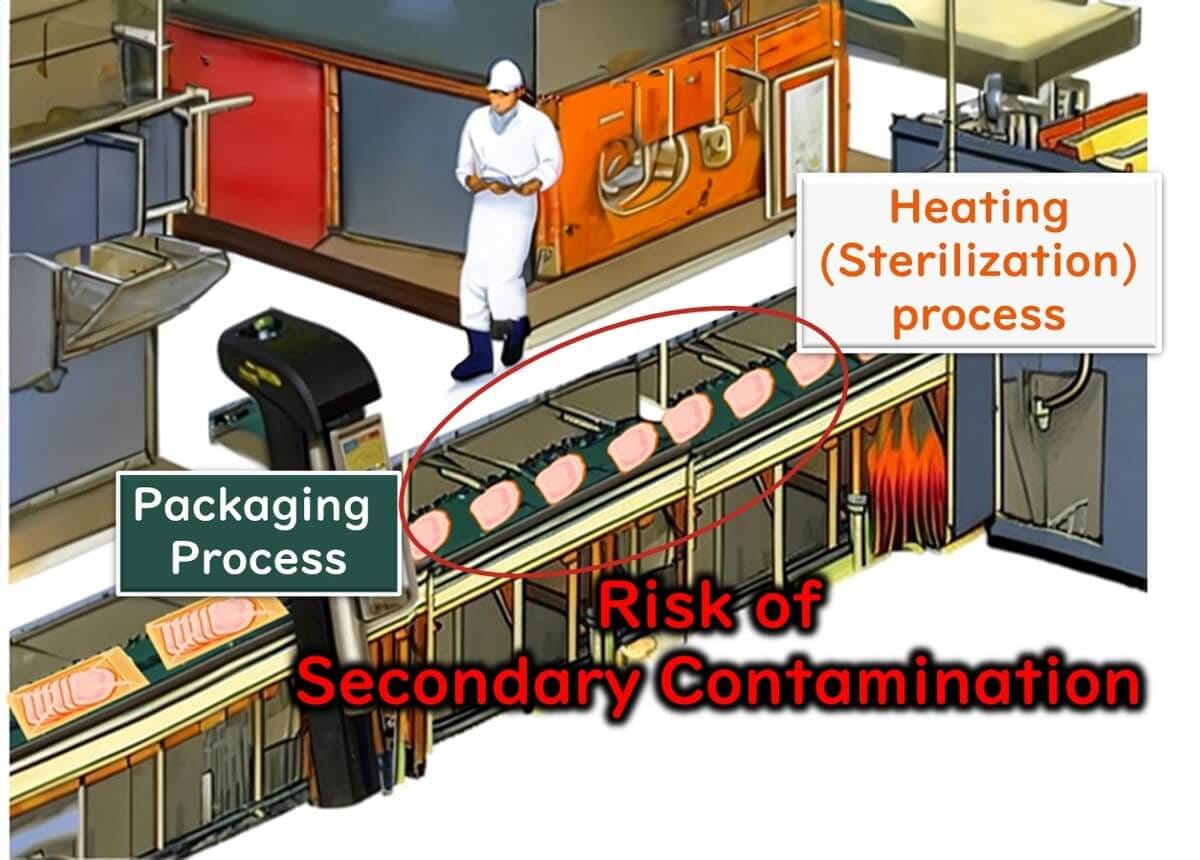
Therefore, as a prerequisite to HACCP, implementing an Environmental Monitoring Programme (EMP) is essential for effective microbiological testing and contamination control.
The Lack of a Standardised Protocol in Environmental Monitoring
Although several guidelines and standards have been published (e.g., USDA FSIS 2014, FDA 2017, ISO 18593:2018 、3M & Cornell University2019, EN 17141:2020), there is currently no single, internationally recognised protocol for environmental monitoring in food manufacturing.

Most documents focus on monitoring pathogenic microorganisms, particularly Listeria monocytogenes and Salmonella. However, few address indicator organisms such as E. coli, and even fewer consider spoilage microbes like Pseudomonas, lactic acid bacteria, moulds, and yeasts.
Monitoring protocols must be tailored to the characteristics of each factory, depending on factors such as:
Type of food produced
Facility layout and design
Scale and automation of operations
Number of employees
Sterilisation processes used
Objectives of Environmental Microbiological Monitoring
The main aims of monitoring in a food factory setting include:
Verifying the effectiveness of cleaning and sanitising procedures
Detecting the presence of specific pathogens (persistent or transient)
Identifying potential contamination sources
Understanding the microbial ecology within the plant environment
Basics of Microbiological Environmental Monitoring
Each environmental monitoring programme should be custom-designed. The following outlines common practices based on international guidelines.
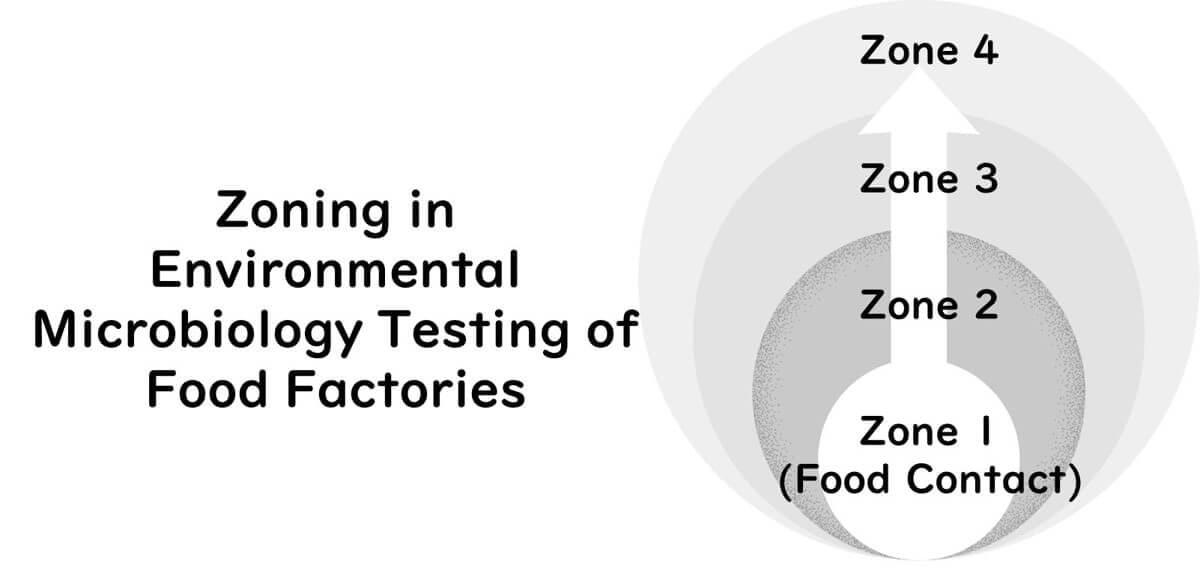
Zoning for Environmental Sampling
Zoning is a key component of environmental monitoring. Sampling areas are commonly divided into three or four zones:
Zone 1: Exposed food contact surfaces (highest risk)
Zone 2: Non-food contact surfaces adjacent to food zones
Zone 3: More distant surfaces within or near the processing area
Zone 4: Areas outside the processing environment
When using a three-zone model, Zones 2 and 3 are combined.
Zone 1 Examples
Conveyor belts transporting unpackaged food
Food handling tables
Internal surfaces of pipes and mixing vessels
Cooling rack covers
Filler nozzles
Storage box interiors
Utensils for food handling

Zone 2 Examples
Walls, ceilings, and floors near exposed food
Equipment parts beyond food contact surfaces
Areas within the same room as Zone 1

Zone 3 Examples
Forklifts, hand trucks, and carts
Walls, floors, and drains near Zone 2

Zone 4 Examples
Locker rooms, toilets, and corridors
Entrances, receiving areas, and warehouses
Preventive contamination control measures should be in place when moving from Zone 4 to Zone 3 and then to production zones.


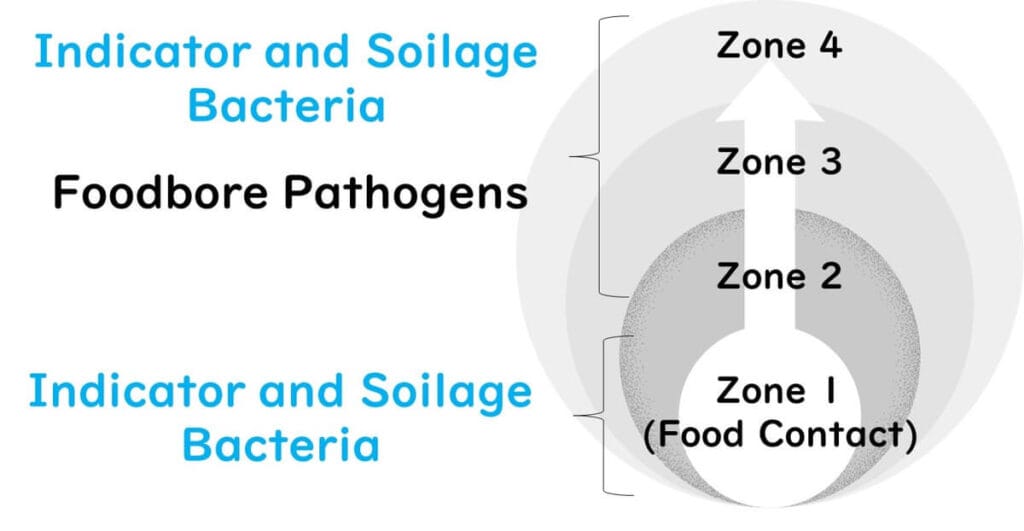
Target Microorganisms for Environmental Monitoring
The organisms targeted vary by zone and purpose:
Zones 1–4: Indicator and spoilage bacteria
Zones 2–4: Indicator bacteria, spoilage bacteria, and pathogens
Pathogens are typically excluded from routine Zone 1 monitoring due to the delay in product release caused by testing. Pathogen monitoring in Zone 1 is reserved for high-risk situations.
Frequently Targeted Pathogens
Listeria monocytogenes
Key concern in ready-to-eat (RTE) foods
Known to form persistent biofilms
Salmonella
Can persist in factory environments
Causes secondary contamination (e.g., in peanut butter, chocolate)
Cronobacter sakazakii
Critical for powdered infant formula
Capable of long-term survival in dry environments
Designing a Sampling Plan
Phase 1: Random Sampling
Initial sampling should be random to ensure all areas are fairly assessed. All food contact surfaces must be included to confirm post-startup hygiene effectiveness.

Phase 2: Combined Random and Targeted Sampling
Using Phase 1 data, Phase 2 incorporates both:
Random sampling
Targeted sampling based on prior results or environmental risk (e.g., drain backflow, condensation, sanitation concerns)
Example: For each production line, 3–5 samples might be taken—1–2 targeted, the rest random.
In areas with known hygiene problems, increase sampling frequency accordingly.

Sampling Frequency Guidelines
Annual and Monthly Planning
Sampling frequency depends on:
Product type and volume
Production frequency
Facility age and design
Shared use of equipment between raw and RTE foods
No Listeria growth: Monthly sampling
Detected growth: Weekly or more frequent sampling
Positive Listeria spp. results: Increase sampling rate
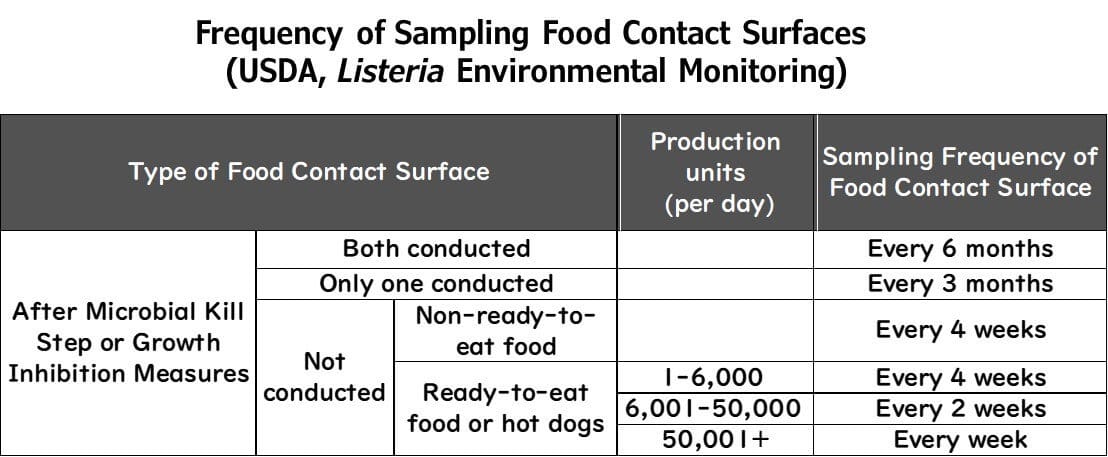
Example: USDA FSIS Recommendations
Sampling Zone 1 depends on whether sterilisation or growth suppression measures are in place.
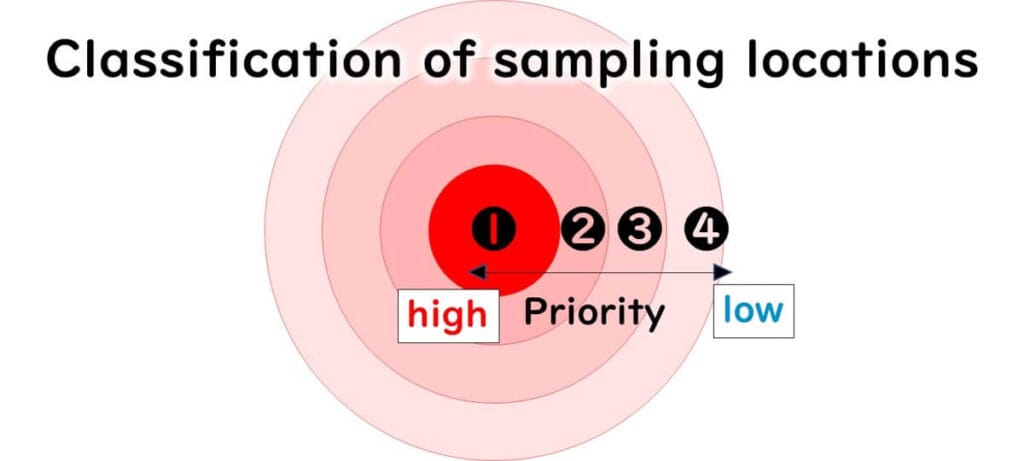
Site Risk Classification
Sampling sites may be categorised as:
High-risk: Always sampled
Rotational: Sampled in turn
Random: Sampled without a fixed plan
Free-choice: Chosen by the sampler
Timing of Daily Sampling
Sampling can occur:
Before production: To verify cleaning/sanitising
During production: For general monitoring
After production: To detect contamination over time

Recommended Practices
Run equipment before sampling to dislodge hidden microbes
ISO 18593:2018 suggests sampling at least two hours after production begins or at the end of a production cycle
If contamination is suspected during production, sample while machinery is operating

Swab Testing Methods and Areas
ISO 18593:2018 recommends:
1,000–3,000 cm² swabbed for detecting specific pathogens
100 cm² swabbed for general microbial counts
USDA FSIS advises sampling the largest possible area, ideally 30 cm x 30 cm. If equipment is small (e.g., buttons), smaller areas are acceptable.

Recommended Reading and References
For those seeking further information, the following resources are recommended:
Environmental monitoring program to support food microbiological safety and quality in food industries: A scoping review of the research and guidelines Food Control,130, 108283 (2021)
Guidance for Environmental Testing for Listeria: USDA FSIS (2014)
Draft Guidance for Industry: Control of Listeria monocytogenes in Ready-To-Eat Foods: FDA (2017)
Microbiology of the Food Chain – Horizontal Methods for Surface Sampling: ISO 18593:2018 (2018)
Environmental Monitoring Handbook for the Food and Beverage Industry: 3M & Cornell University (2019)
Environmental Monitoring Program to Support Food Microbiological Safety and Quality in Food Industries: A Scoping Review of the Research and Guidelines: Food Control, 130, 108283 (2021)
Conclusion and Upcoming Case Studies
This article has outlined the foundational concepts of microbiological environmental monitoring in food factory hygiene management. However, theory alone is not sufficient—real-world case studies provide practical insights into applying these principles effectively.
In future blog posts, we will introduce individual monitoring case studies based on actual factory scenarios. These will be presented intermittently and aim to deepen practical understanding of microbiological monitoring for hygiene control.
Related posts:
The Role of Microbial Testing in HACCP (3) While HACCP aims to reduce reliance on end-product testing, microbial...
Understanding the Accuracy of Microbial Testing of Food (3) To ensure food safety, it's essential to grasp the purpose...
Easy-to-Understand Food Microbiology Testing Sampling Plans in the ICMSF (3) The meaning of a "negative" result in food microbiological testing...
How HACCP Reduces Reliance on Microbial Testing Through Process Control (3) HACCP (Hazard Analysis Critical Control Point) is a system designed...

Discover expert-led lessons in food microbiology designed for professionals and beginners alike.

Author of this Blog: Bon Kimura
Bon Kimura, Professor Emeritus at Tokyo University of Marine Science and Technology (TUMSAT), specializes in food microbiology. He obtained his PhD from Kyoto University and became a professor at TUMSAT in 2006, serving as Dean of the Faculty of Marine Science from 2012-2015. Kimura has published over 200 international papers on food safety, pathogens, and spoilage bacteria. He has received multiple awards, including the Japanese Society for Food Microbiology Award (2019). Kimura also served as an editor for the International Journal of Food Microbiology from 2012 to 2024, where he was the principal reviewer for 1,927 papers.

Copyright © Introduction to Food Microbiology and Safety All Rights Reserved.